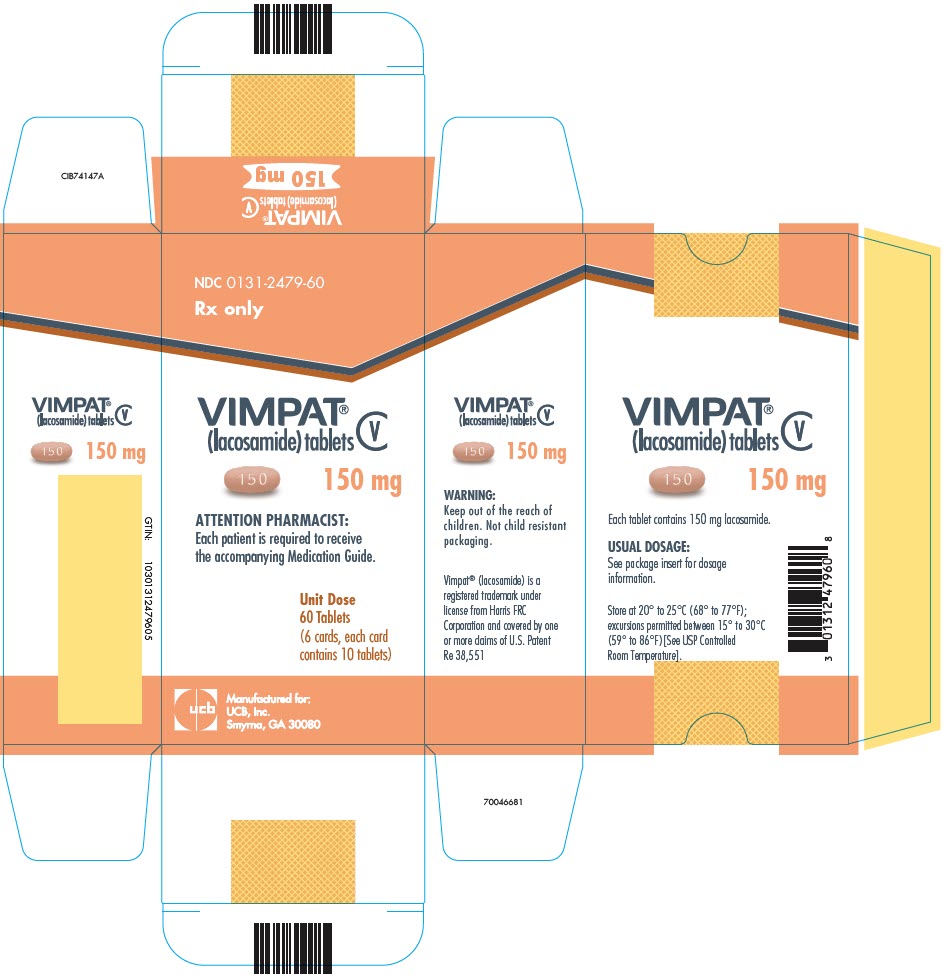
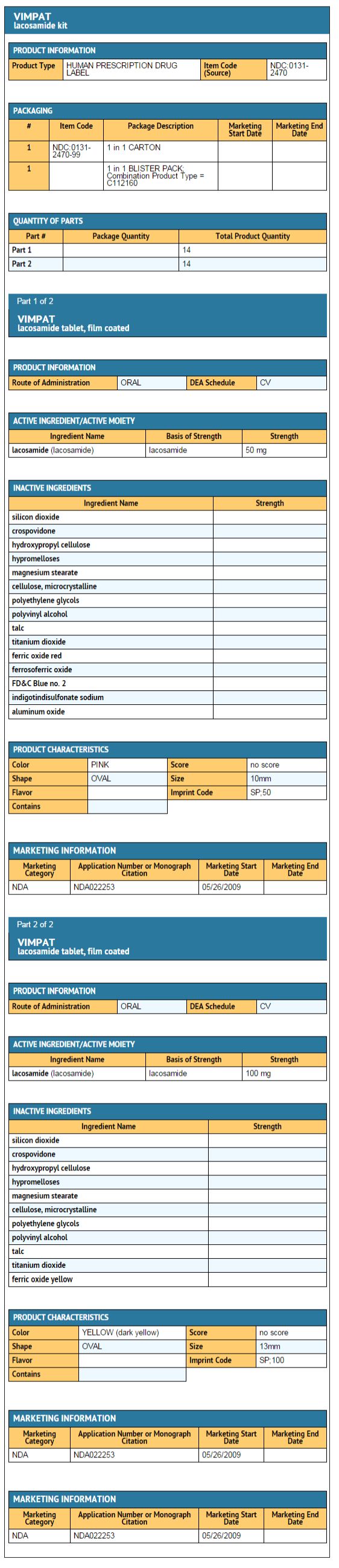
UCB's VIMPAT® (lacosamide) CV now approved by FDA for primary generalized tonic-clonic seizures and expanded pediatric use for people living with epilepsy

These highlights do not include all the information needed to use LACOSAMIDE INJECTION safely and effectively. See full prescribing information for LACOSAMIDE INJECTION. LACOSAMIDE injection, for intravenous use, CV Initial U.S. Approval:

Neurological adverse events of new generation sodium blocker antiepileptic drugs. Meta-analysis of randomized, double-blinded studies with eslicarbazepine acetate, lacosamide and oxcarbazepine - ScienceDirect

Effect of lacosamide on ethanol induced conditioned place preference and withdrawal associated behavior in mice: Possible contribution of hippocampal CRMP-2 - ScienceDirect








:max_bytes(150000):strip_icc()/lamictal-withdrawal-symptoms-380259_v2-8c92ee1d1cf7493199fad1ba24c0da45.png)